Subjects
Grades
The periodic table groups all chemical elements according to certain characteristics.
Each square of the periodic table corresponds to a chemical element. These squares contain important information about the element, such as:
its atomic number
its name
its chemical symbol
its atomic mass
The colour of the square also generally gives information about the state of the element.
The information can be located in different parts of the square, depending on the periodic table. It is important to consult the legend of the table to find your way around.
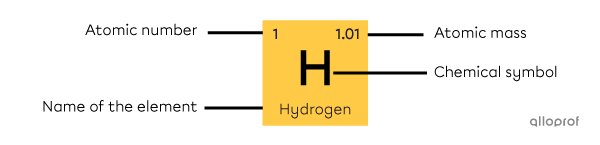
A square in the periodic table
Each element has its own number, known as its atomic number. The 118 elements of the periodic table are classified in ascending order of atomic number, from 1 to 118. The atomic number is assigned to each element according to the internal structure of its atoms.

Atomic Number
In addition to having a unique atomic number, each element has a unique name, which is used to identify it in common vocabulary. The origin of these names varies greatly. Some names are given in honour of deities, scientists or places. For example, curium is named after twice Nobel Prize winner Marie Curie. Other names are given according to the properties of the elements. This is the case with iodine, which comes from the Greek word iodes, meaning violet, which is the colour of pure iodine[2]. The name of the element can be different in different languages.

Name of the element
The chemical symbol consists of an uppercase letter, sometimes accompanied by a lowercase letter. The chemical symbol is derived from the Latin name of the element. It is the same in all languages. Often, it also corresponds to the first letters of the name in English, but this is not always the case. Hydrogen (H), helium (He), lithium (Li) and carbon (C) are examples of elements whose symbol corresponds to the first letters of their name. However, the word silver comes from the Old English word siolfur[3]. Silver's chemical symbol is Ag.
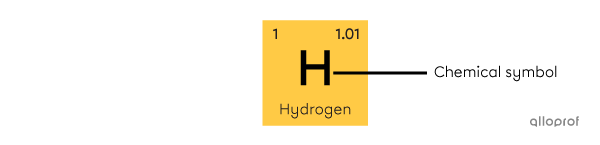
Chemical symbol
The atomic mass of an element is the mass of a single atom of that element. Since the mass of an atom is extremely small, it is generally measured in atomic units (u) to simplify calculations. This is the unit used in the periodic table. One atomic unit corresponds roughly to the mass of hydrogen, the lightest chemical element.

Atomic mass
The colour of a square in the periodic table generally indicates the state of the substance at a temperature of |0^\circ\text{C}.| The colours and the state to which they correspond are identified in a legend. In the periodic table at the top of this page, the legend indicates that the yellow squares correspond to elements in gaseous state, blue squares correspond to elements in liquid state, green squares correspond to elements in solid state and orange squares correspond to elements that are synthetic solids. Synthetic solids do not exist in nature; they are produced in laboratories.
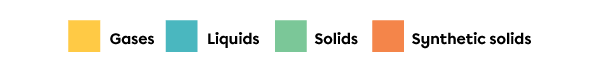
Example of a colour legend for the squares of a periodic table