Subjects
Grades
Matter makes up all substances that have a mass and occupy space.
Matter consists of small particles, called atoms, that are invisible to the naked eye. There are different types of atoms and they can exist on their own or bond with other atoms.

Moments in the video:
An atom is a particle of matter invisible to the naked eye. It is a building unit of molecules. To find out more about atoms, check out this concept sheet:
A molecule is a group of two or more atoms of the same or different types chemically bonded together. To find out more about molecules, check out this concept sheet:
An element corresponds to one type of atom. The elements are organized in the periodic table of elements. To find out more about the periodic table of the elements, check out this concept sheet:
Atoms, molecules and elements can be seen in the images of the following substances.
In this image of methane, the following particles can be identified:
There are 5 atoms: 1 atom of carbon (C) and 4 atoms of hydrogen (H).
There is 1 molecule because it is a group of several atoms bonded together.
There are 2 elements because there are 2 types of atoms: hydrogen (H) and carbon (C).
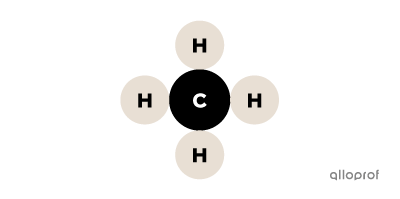
Methane
In this image of nitrogen, the following particles can be identified:
There are 2 atoms: 2 atoms of nitrogen (N).
There is 1 molecule because it is a group of several atoms bonded together.
There is 1 element because there is only 1 type of atom: nitrogen (N).

Nitrogen