Subjects
Grades
The pH, short for potential of hydrogen, is a measure used to determine the acidity, neutrality or basicity (alkalinity) of a solution. The pH has no unit of measurement.
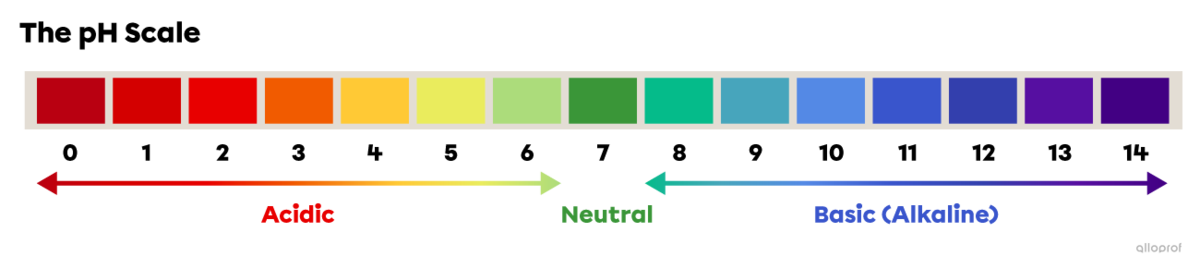
The pH scale is used to compare the acidity, neutrality or basicity (alkalinity) of solutions. The pH scale generally ranges from 0 to 14.
On the pH scale, 0 is the most acidic value, 7 is neutral and 14 is the most basic, or alkaline, value.
Acid-base indicators, commonly referred to as pH indicators, are substances that change colours depending on the pH.
Some of the application of acid-base indicators in laboratories are:
To determine the acidity, neutrality or basicity (alkalinity) of a solution.
To determine the pH value of a solution.
The following image shows the colours of acid-base indicators depending on the pH.
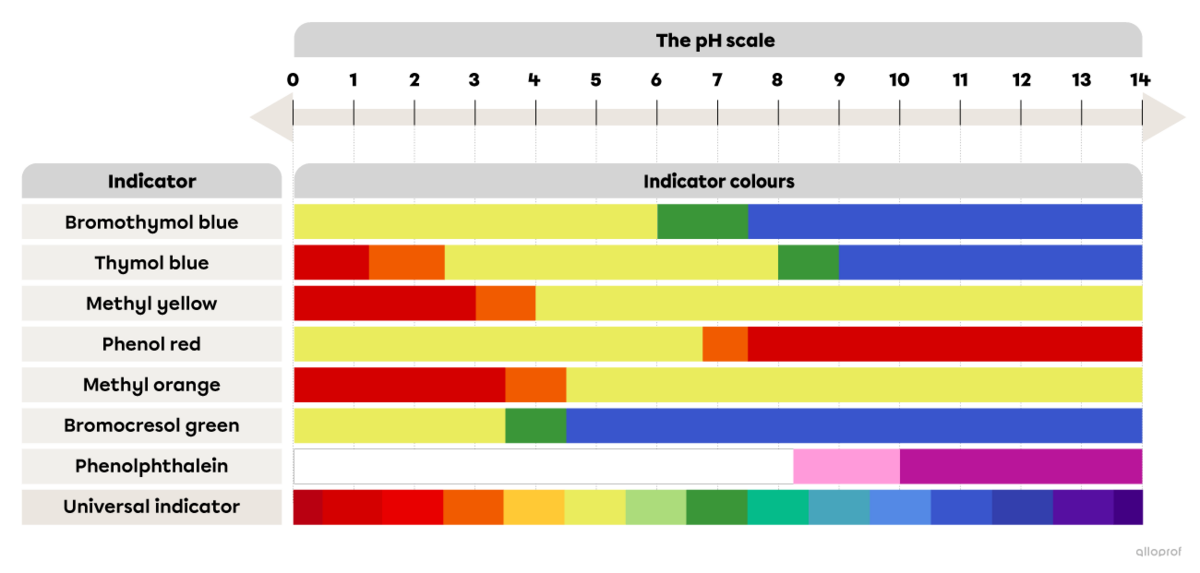
The colour range of some acid-base indicators depending on the pH
Phenolphthalein is an indicator that can be used to determine if the solution is basic. When a few drops of phenolphthalein are added to the solution in question, a colour change may occur.
If phenolphthalein remains colourless, the solution is either acidic (pH below 7), neutral (pH 7), or basic (pH 7-8.2).
If the indicator turns pink, the solution is basic with a pH between 8.2 and 10.
If the indicator turns fuchsia, the solution is basic with a pH above 10.
In short, if phenolphthalein turns pink or fuchsia when mixed with a solution, the solution is definitely basic.
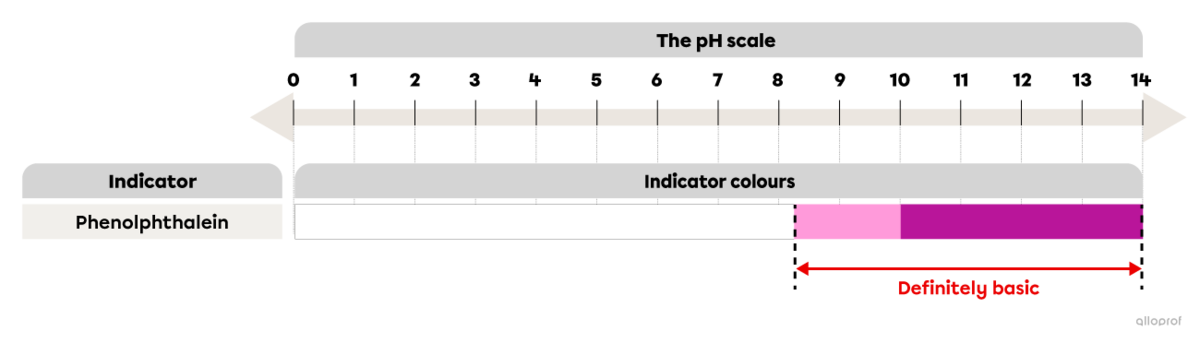
The phenolphthalein colour depending on the pH
A few drops of an acid-base indicator thymol blue are added to a test tube filled with lemon juice (pH 2). Thymol blue is thoroughly mixed with lemon juice and a colour change occurs.
Referring to the image The colour range of some acid-base indicators depending on the pH, determine the colour of thymol blue after it is mixed with lemon juice.

Adding thymol blue to lemon juice
A few drops of methyl yellow are added to an unidentified substance and the solution turns orange. In a second test tube, bromocresol green is added to the same unidentified substance and it turns green.
Referring to the image The colour range of some acid-base indicators depending on the pH, determine the pH interval of the unidentified substance.
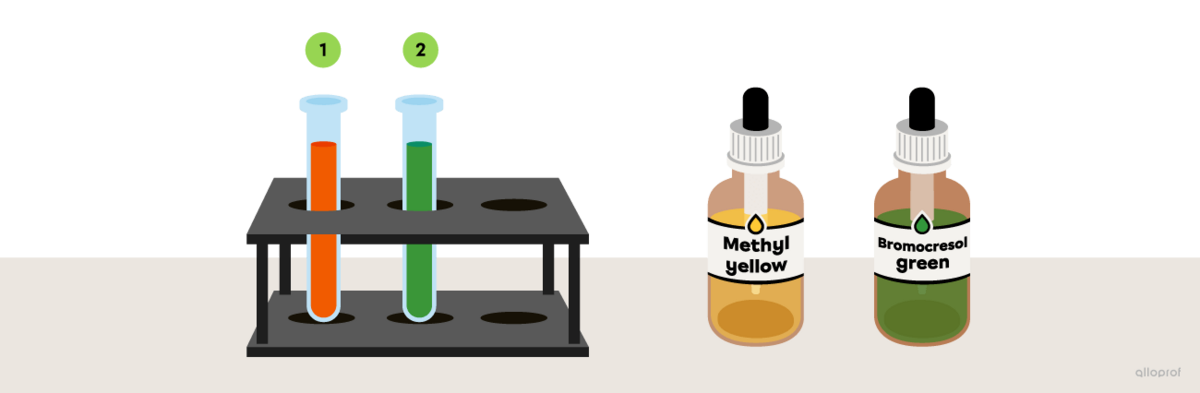
The reaction of methyl yellow and bromocresol green with an unidentified substance