Subjects
Grades
A chemical reaction, sometimes called a chemical change or chemical transformation, occurs when one or more substances, the reactants, interact to form one or more new substances, the products.
A reactant is a substance that is used up during a chemical reaction. Its amount decreases.
A product is a substance formed during a chemical reaction. Its amount increases.
Unlike physical changes, a chemical reaction changes the nature of the substances involved. The reactants of a chemical reaction do not have the same characteristic properties as the products formed.
In addition, a chemical reaction can be recognized by certain indicators, such as a change in colour, the release of gas, the formation of a precipitate, etc.
Chemical reactions can be represented in different ways and grouped into categories referred to as the chemical reaction types.
Different representations can be used to describe the substances involved in a chemical reaction. A chemical reaction is usually represented using the particle model or a chemical equation.
When representing a chemical reaction using the particle model, atoms are usually represented by balls. The colour and size of the balls vary, allowing atoms of different elements to be represented differently. In addition, the balls can be combined to represent molecules and compounds.
The chemical reaction of carbon combustion can be represented using the particle model as follows. In this reaction, 1 atom of carbon reacts with 1 molecule of oxygen to produce 1 molecule of carbon dioxide.
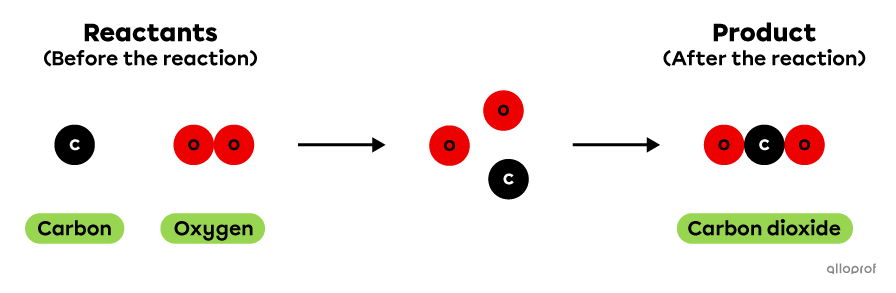
Carbon combustion according to the particle model
A chemical reaction can fit into several types of reactions at once. For example, cellular respiration is both an oxidation and a decomposition reaction.
A synthesis reaction is a chemical reaction where two or more reactants interact to form a new product.
A synthesis reaction can be recognized by analyzing the number of different reactants and products, as well as the complexity of the substances involved.
It is possible to recognize a synthesis reaction by the following clues:
There are more reactants than products.
The reactants are simpler substances than the product(s).
In the following reaction, ethylene reacts with hydrogen to form ethane.
This is a synthesis reaction because two reactants, ethylene and hydrogen, react to form one product, ethane. There are therefore more reactants than products. Furthermore, the two reactants are simpler molecules than the product: ethylene and hydrogen each contain fewer atoms than ethane.
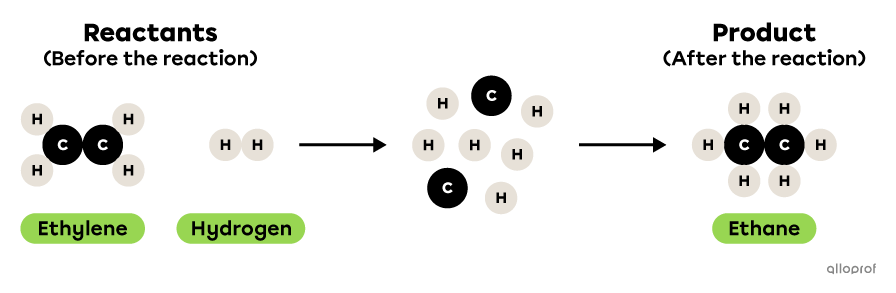
Ethane synthesis
Photosynthesis is a synthesis reaction where carbon dioxide reacts with water in the presence of solar energy to produce glucose and oxygen.
It can be recognized as a synthesis reaction because the glucose molecule produced is more complex than the two reactants involved.
In plants, this chemical reaction takes place in the chloroplasts.
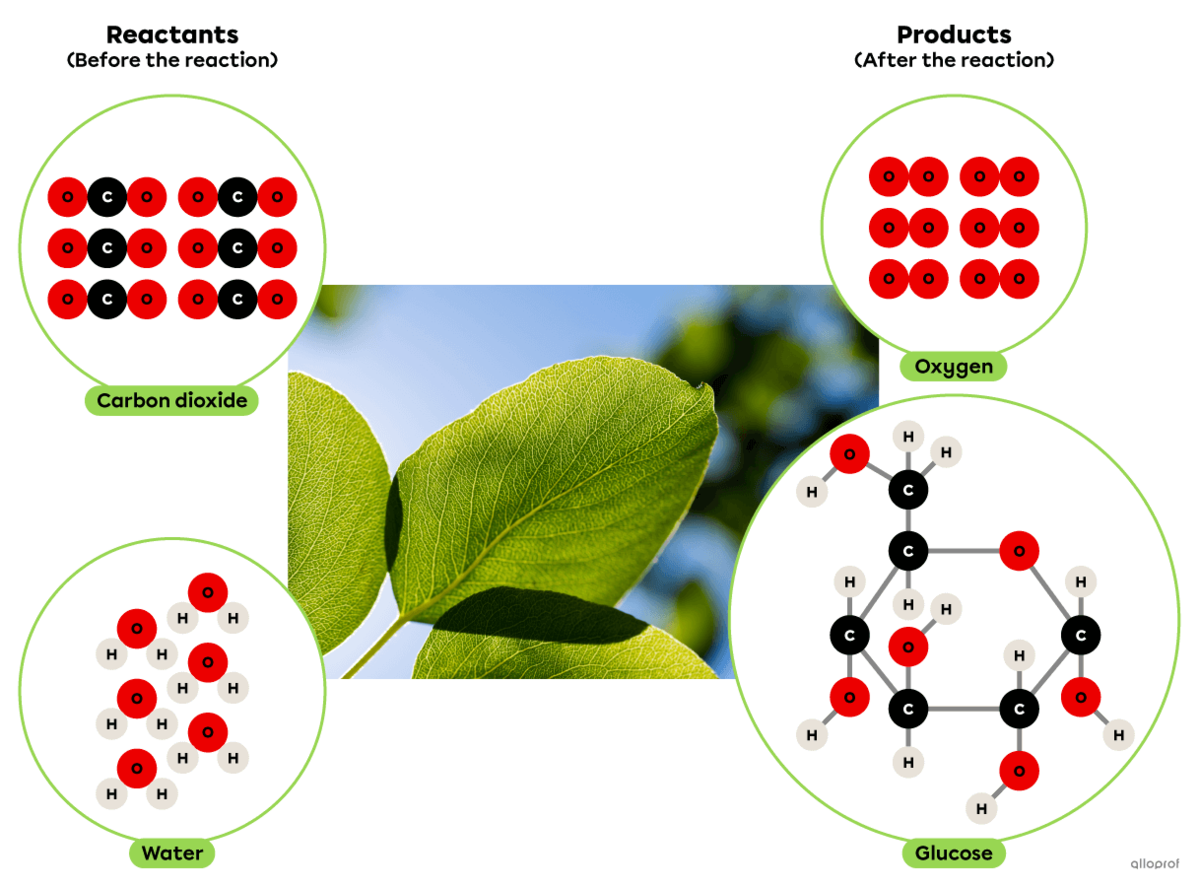
Photosynthesis according to the particle model
A decomposition reaction is a type of chemical reaction where the atoms of a reactant dissociate and rearrange to form several products.
A decomposition reaction can be recognized by analyzing the number of different reactants and products, as well as the complexity of the substances involved.
It is possible to recognize a decomposition reaction by the following clues:
There are more products than reactants.
The product(s) formed are simpler substances than the reactant(s).
In the following reaction, hydrogen peroxide decomposes to form hydrogen and oxygen.
It is recognized as a decomposition reaction because only one reactant, hydrogen peroxide, is decomposed to form two products, hydrogen and oxygen. In addition, hydrogen peroxide is a more complex molecule than either of the two products: it contains more atoms than either hydrogen or oxygen.
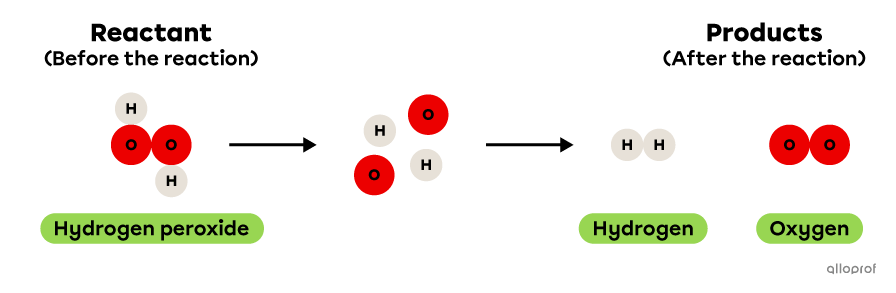
Decomposition of hydrogen peroxide
During chemical digestion, decomposition reactions allow nutrients, which are complex molecules, to be broken down into simpler molecules that can be absorbed by the body.
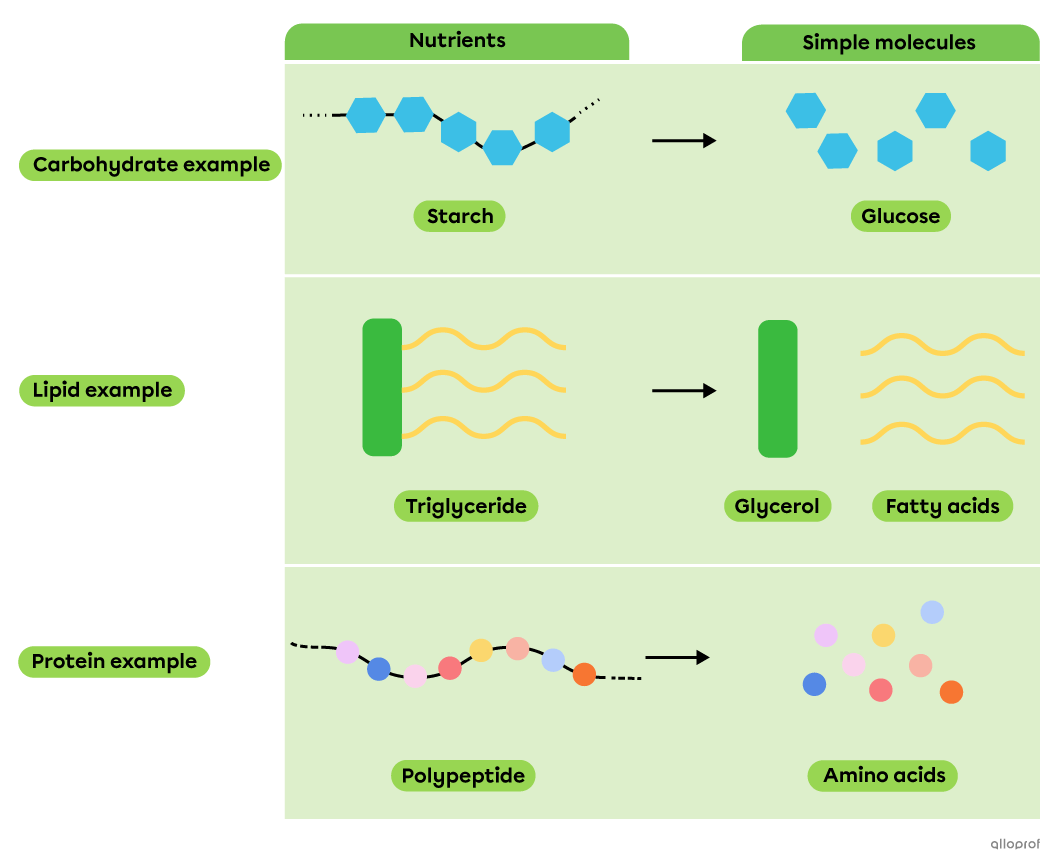
Decomposition of nutrients into simple molecules
A precipitation reaction is a chemical reaction that occurs when two substances in a solution interact to produce an insoluble or slightly soluble solid called a precipitate.
When a sodium chloride solution is mixed with a silver nitrate solution, a whitish precipitate is formed.
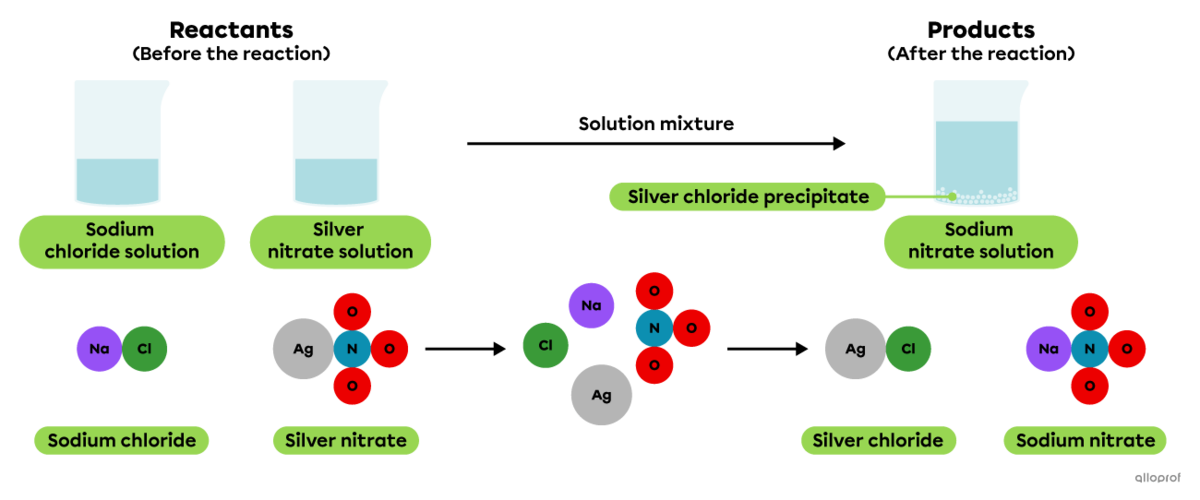
Precipitation of silver chloride
An oxidation reaction is a chemical reaction involving oxygen or a substance with oxygen-like chemical properties among the reactants.
In the following reaction, iron reacts with oxygen to form iron oxide, commonly known as rust. This is an oxidation reaction because oxygen is one of the reactants involved in the reaction.

Oxidation of iron
A combustion reaction is a chemical reaction where an oxidation reaction occurs and energy is released.
In the following combustion reaction, propane reacts with oxygen to form carbon dioxide and water. In addition, the combustion of propane results in the release of thermal energy which is commonly used for cooking or heating buildings.
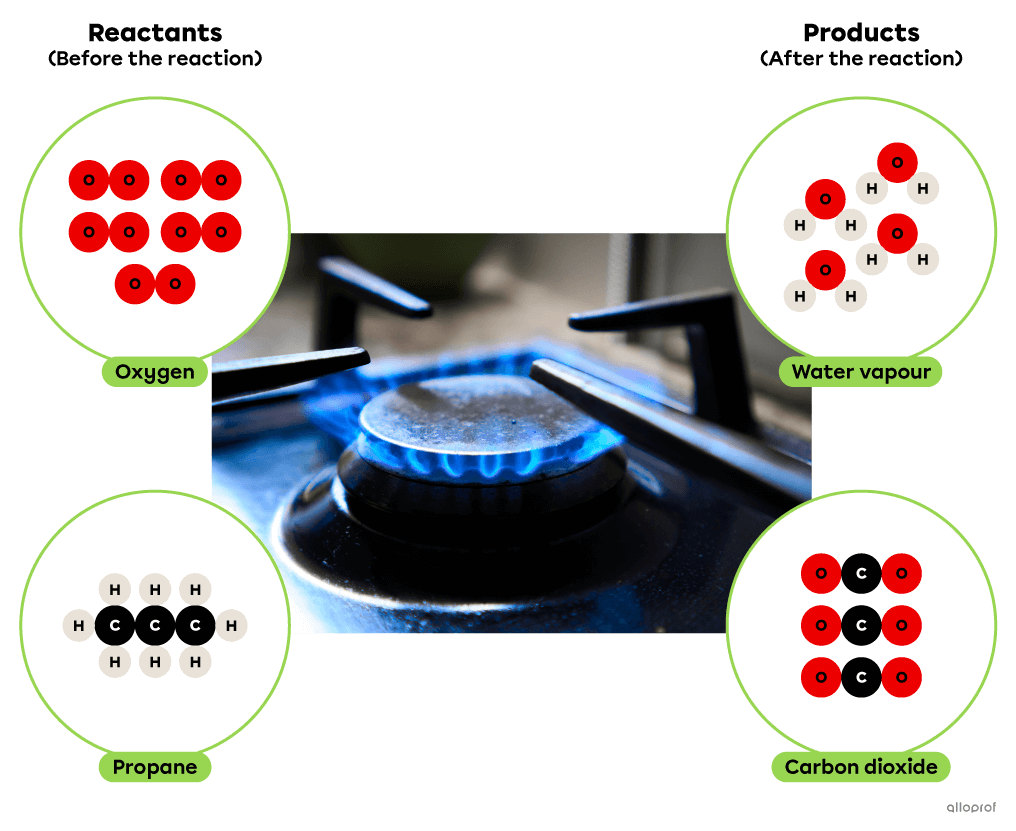
Combustion of propane
Cellular respiration is a chemical reaction that takes place in animal and plant cells.
It is an oxidation reaction where glucose reacts with oxygen to form carbon dioxide and water vapour. In addition, cellular respiration produces the energy essential for the proper functioning of cells.
Since it involves an oxidation reaction and the release of energy, it can also be said that cellular respiration is a combustion reaction.
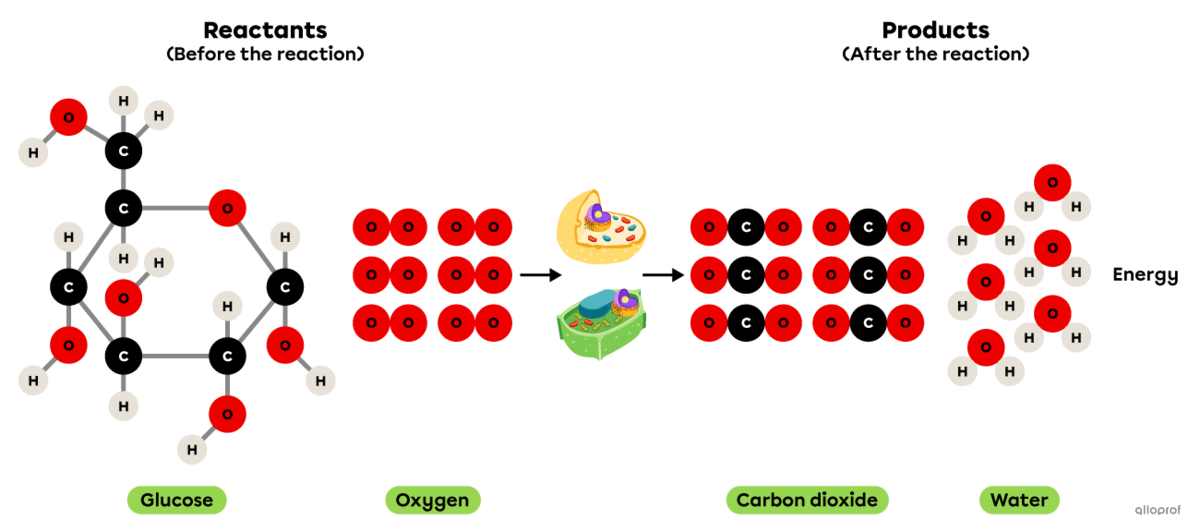
Cellular respiration