Subjects
Grades
A molecule is a group of at least two chemically bonded non-metals.
When metals combine with non-metals, they form salts and not molecules. Salts are also called ionic compounds.
A molecule can be identified using its chemical formula or its name.
The chemical formula of a molecule is specifically referred to as the molecular formula. It contains the chemical symbols of all the atoms that make up the molecule. Each chemical symbol is accompanied by a subscript if there is more than one atom of this type present.
It is important to respect nomenclature rules when naming molecules.
The following molecule can be identified either by its common name, methane, or by its chemical formula, |\text{CH}_4.|
The |\text{C}| symbol indicates the molecule contains carbon.
The lack of subscript next to |\text{C}| means that the molecule only has one atom of carbon.
The |\text{H}| symbol indicates the molecule contains hydrogen.
The subscript 4 that follows the |\text{H}| means that there are 4 hydrogen atoms in the molecule.

A molecule can be represented using Dalton’s atomic model.
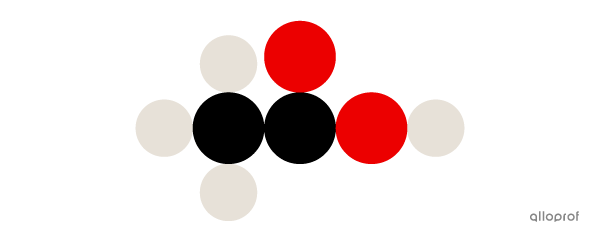
Here, a molecule of acetic acid |(\text{CH}_3\text{COOH})| is represented according to Dalton’s atomic model.
A molecule can also be represented according to Lewis notation and the Rutherford atomic model. Both topics are covered in the Environmental Science and Technology (EST) course.
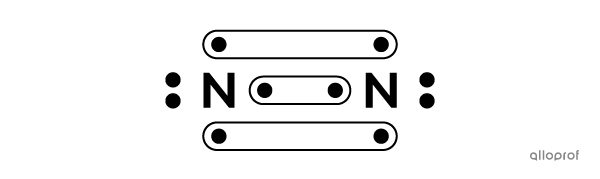
Here, molecular nitrogen |(\text{N}_2)| is represented using Lewis notation.
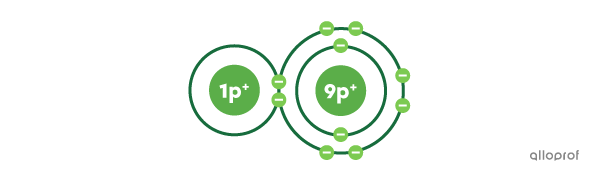
Here, a molecule of hydrofluoric acid |(\text{HF})| is represented using Rutherford-Bohr’s atomic model.