Subjects
Grades
A molecule is a group of at least two chemically bonded atoms.
According to Dalton’s atomic model, atoms are represented by coloured balls. Therefore, a molecule is represented by a combination of at least 2 balls.
The molecule represented by the following model is a combination of three chemically bonded atoms.
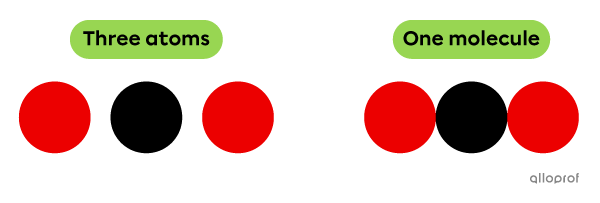
Atoms and a molecule
The periodic table of elements is where all elements are found, meaning all types of atoms. A molecule is a combination of at least two atoms that are found in the periodic table. They can be the same type or different types.
A molecule of oxygen |\text{(O}_2)| is made of two atoms of oxygen |(\text{O}).|

Oxygen molecule
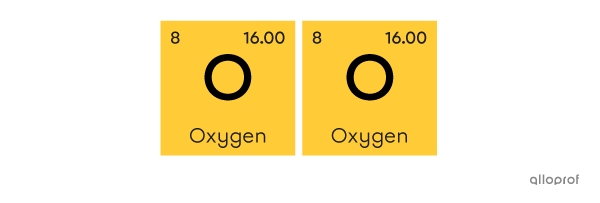
Two atoms of oxygen
A molecule of acetic acid |(\text{CH}_3\text{COOH})| is made of four atoms of hydrogen |(\text{H}),| two atoms of carbon |(\text{C})| and two atoms of oxygen |(\text{O}).|
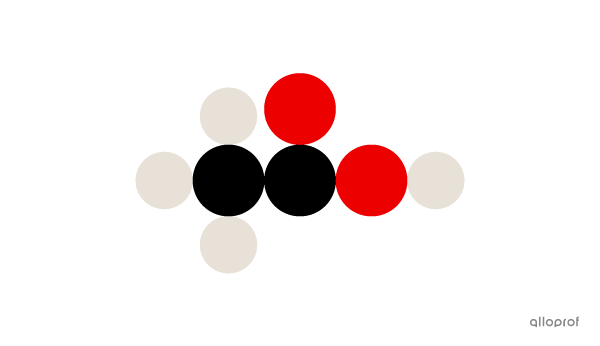
An acetic acid molecule
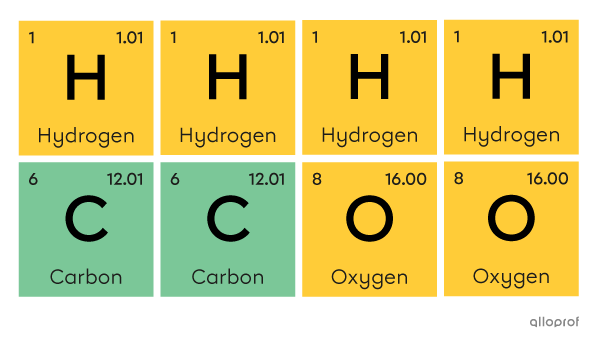
Four hydrogen atoms, two carbon atoms and two oxygen atoms
In order to recognize a molecule using its chemical formula, look at the types of capital letters and/or subscripts.
Every type of atom (element) has a unique symbol that contains only one capital letter. Therefore, a chemical formula that contains several capital letters indicates that there are several types of atoms. For example, |\text{HCl}| contains 2 capital letters. Therefore, this substance contains two types of atoms: hydrogen and chlorine. It’s a molecule.
In a chemical formula, a subscript indicates the number of atoms of a given type. The subscripts are written only when more than one atom of the same type is included. Remember, when there is a subscript, there is more than one atom. For example, |\text{I}_2| has a subscript of |2.| This substance contains two atoms of iodine |(\text{I}).| It is a molecule.
A molecule does not have the same properties as the elements that compose it.
Once the chemically bonded atoms separate and rearrange their bonds in a different way, they form new molecules. These new molecules do not have the same properties as the initial molecules despite the fact that they are made of the same atoms and a chemical reaction occurred.
A water molecule is formed from oxygen |(\text{O})| and hydrogen |(\text{H}).|
Molecular oxygen |(\text{O}_2)| made of oxygen atoms |(\text{O})| is a gas essential for respiration. Molecular hydrogen |(\text{H}_2)| made of hydrogen atoms |(\text{H})| is a combustible gas.
The water molecule |(\text{H}_2\text{O})| itself is neither essential for respiration nor combustible. Instead, one of its main properties is its ability to dissolve many substances, such as salt, sugar and carbon gas, for example.
Even if the molecule |\text{H}_2\text{O}| is formed by atoms that are found in the |\text{O}_2| molecule and in the |\text{H}_2| molecule, the properties of these three molecules are very different.
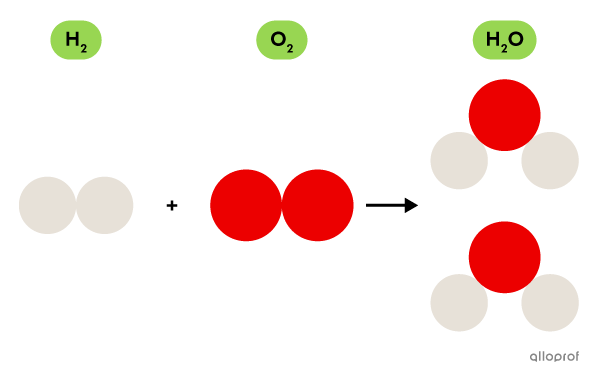
Formation of water molecules